 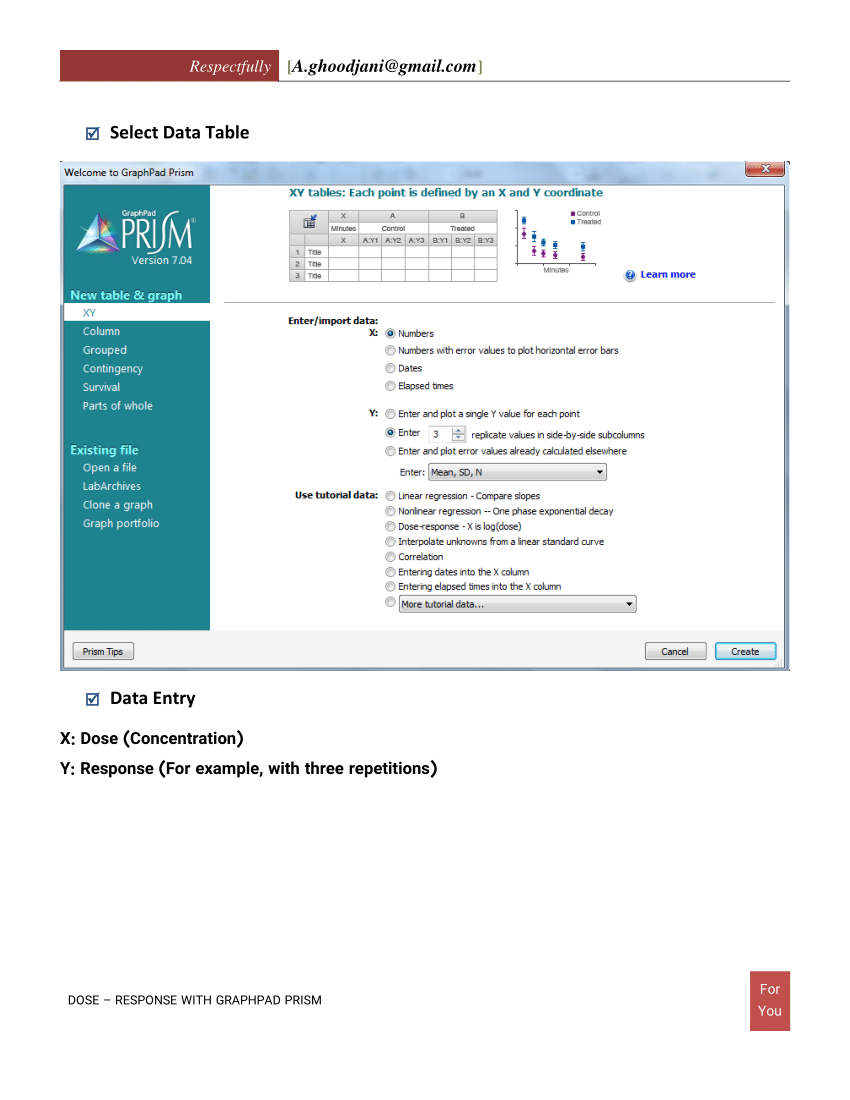
How to calculate IC50 using graphpad prism? I have done serial dilution for a drug having range of 100 nM, 33 nM, 10 nM, 3 nM, 1 nM, 0.3 nM, 0.1 nM, 0.03 nM, 0.01 nM and control (having no drug) Navigation: REGRESSION WITH PRISM 9 > Nonlinear regression with Prism > Models (equations) built-in to Prism > Dose-response - Key concepts. Subscribe to: Post Comments (Atom) Bermain Biola Biola merupakan alat musik gesek yang bunyinya benar-benar menghanyutkan jika dimainkan oleh orang yang pintar menggeseknya. GraphPad Prism 8.4.3.686 Crack with Serial Number 2020 Free Download. The right way to average IC50 data is to use the geometric mean because you're working with an exponential value, which turns out to be 3.7 mM (after skipping the complicated math) When reporting the IC50 values of a curve dose-response obtained with Prism-(Non linear regression, log conc-normalized response), do I need to report the SEM? If yes, can I calculate it from. If you calculate the arithmetic mean, you get an average IC50 of 3.5 mM, but that is incorrect. The biological component could be an enzyme. drug) is needed to inhibit, in vitro, a given biological process or biological component by 50%. IC 50 is a quantitative measure that indicates how much of a particular inhibitory substance (e.g. The half maximal inhibitory concentration (IC 50) is a measure of the potency of a substance in inhibiting a specific biological or biochemical function. Comparative analysis of IC90 values showed that extracts from four plants (Cymbopogon spp, Moringa oleifera, Vernonia amygdalina and Allium sativum) had IC90 values less than 200 g/ml, five (Azadirachta indica, Khaya senegalensis, Carica papaya, Eucalyptus spp and Aloe vera. IC50 and IC90 values were extrapolated from two graphical approaches. Top and Bottom are plateaus in the units of the. A Hill slope greater than 1.0 is steeper, and a Hill slope less than 1.0 is shallower. A HillSlope of 1.0 is standard, and you should consider constraining the Hill Slope to a constant value of 1.0. HillSlope describes the steepness of the family of curves. ,graph pad prism,ic50 calculator from AAT. 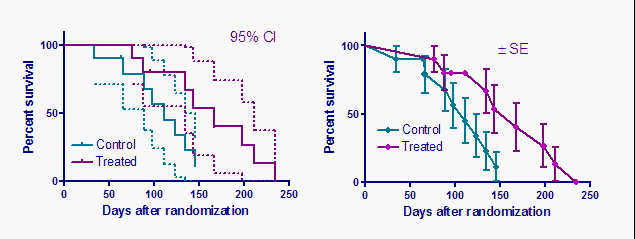
But do not how to do it using GaraphPad Prism- any suggestions To calculate the IC50 value, from the equation of the graph that you got (Y = MX-C), change Y to 50 (50= MX-C) then make X the subject of the formula. Use the link below to download the data for training : I am doing a cyp450 inhibition assay using HTP-fluorescence based assay. response - Variable slope (four parameters). On the Nonlinear regression dialog, open the Dose-Response - Inhibition family of equations, and choose log(inhibitor) vs. 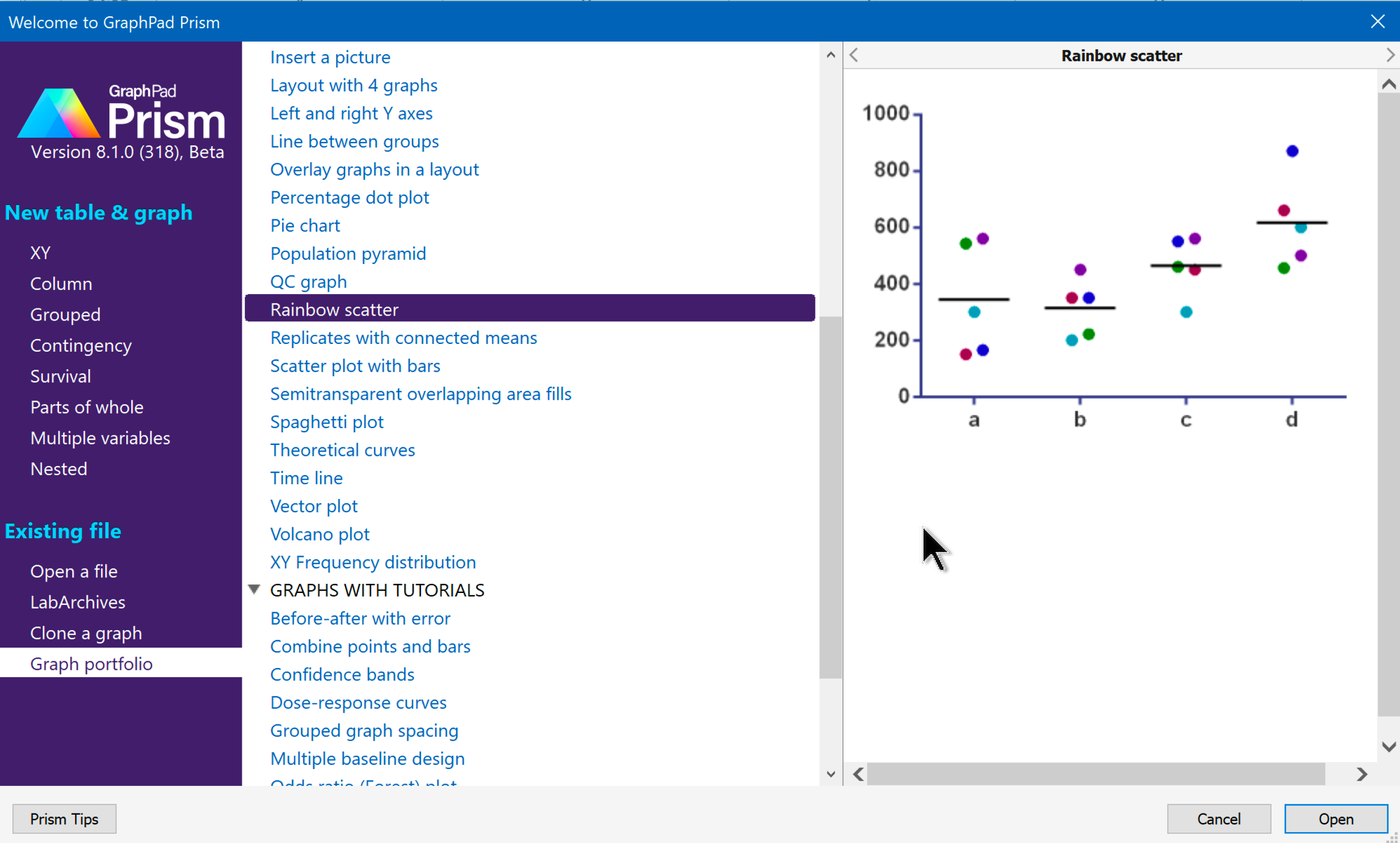
Or click the Nonlinear Regression shortcut button just above the Analyze button. Click Analyze and then Nonlinear regression. 
I this tutorial I demonstrate how to plot an IC50 curve for drug assay data in Graphpad prism 6.I will like to point out an error i made in the video (skip t.Ĩ GraphPad Curve Fitting Guid Prism can interpolate these X values. without the quotes In this tutorial I demonstrate how to find the concentration of a compound that produces any level of Stimulation (EC anything) or Inhibition (IC anything) b. Choose your desired EC Calculate the EC Enter '80' or '90'. Slope factor = (If you don't know, enter '1.0'.) 3. You can also apply this same concept to extrapolate any value from any. Thanks in advance for any guidance, ideas, and/or discussion.This is an alternative method for determining IC and EC anything in Graphpad prism 6. My question is: How do I take the ‘intrinsic activity’ of the enzyme into account (if I need to at all)? Can anyone recommend an equation and appropriate software that can do this? We currently have SigmaPlot, GraphPad, and Enzfitter (for the double substrate inhibition model). We’ve run a full course of analyses, varying the NADH as well as the substrate and we're now trying to fit the data to an appropriate equation. The intrinsic activity introduces a baseline velocity and is complicating our kinetic analyses a bit. Complicating it even further is a noted double substrate inhibition pattern (apparently not uncommon with ping-pong mechanism enzymes). Of course, upon addition of substrate, the velocity of the reaction increases and NADH is consumed more rapidly. I’m working with a rather complicated ping pong, bi-bi double displacement reductase enzyme that oxidizes NADH and then reduces a substrate. Interestingly, the enzyme has intrinsic NADH oxidase activity in the absence of the substrate. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
